Precision RNA Medicines Built to Scale
Precision RNA Medicines Built to Scale
Unlocking Extrahepatic
RNA Therapeutics
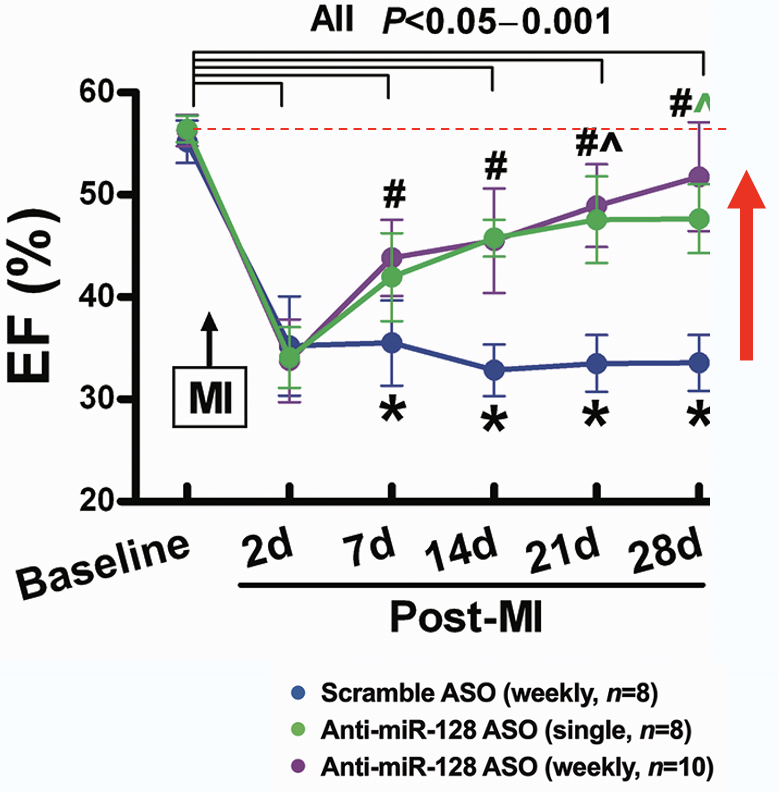
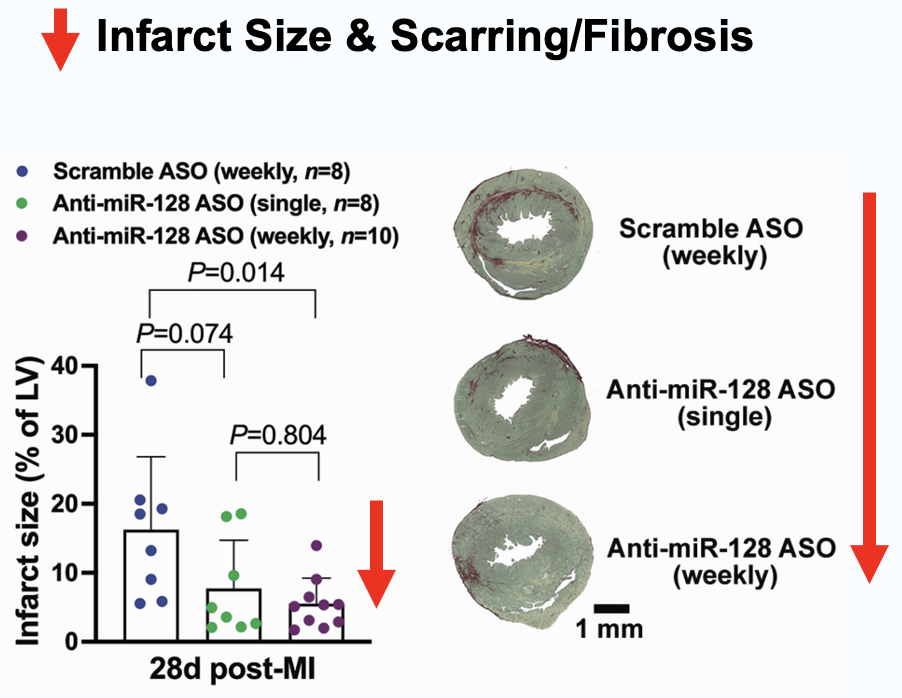
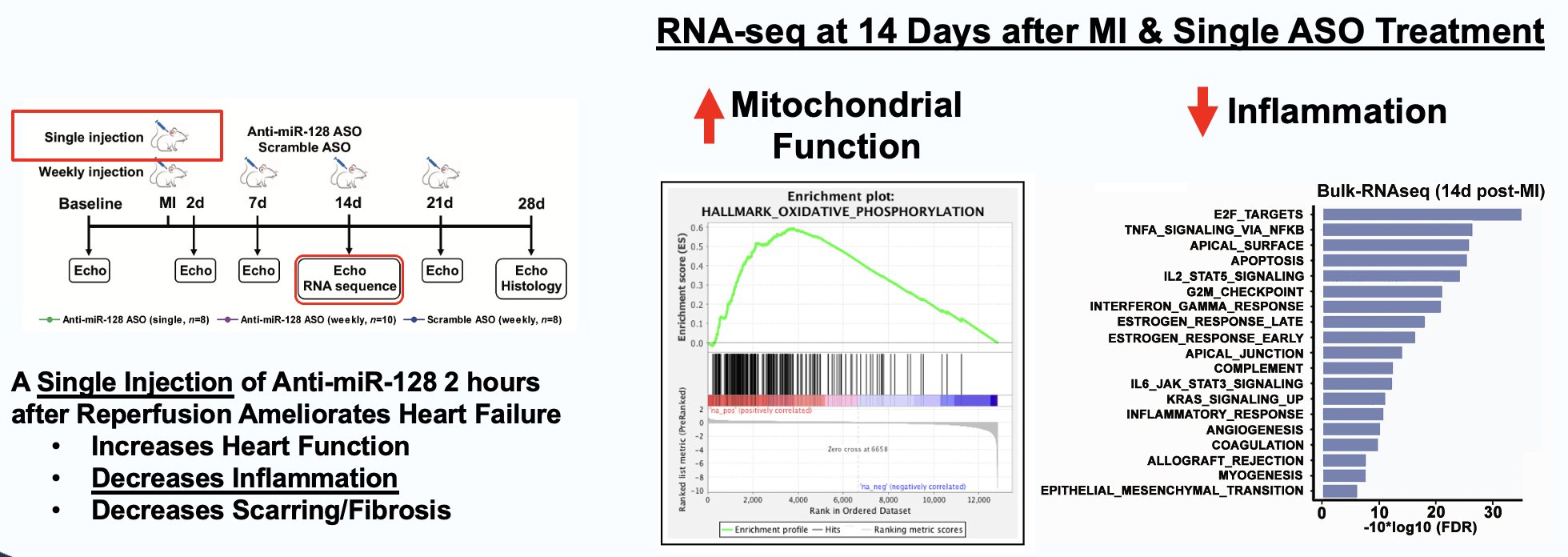
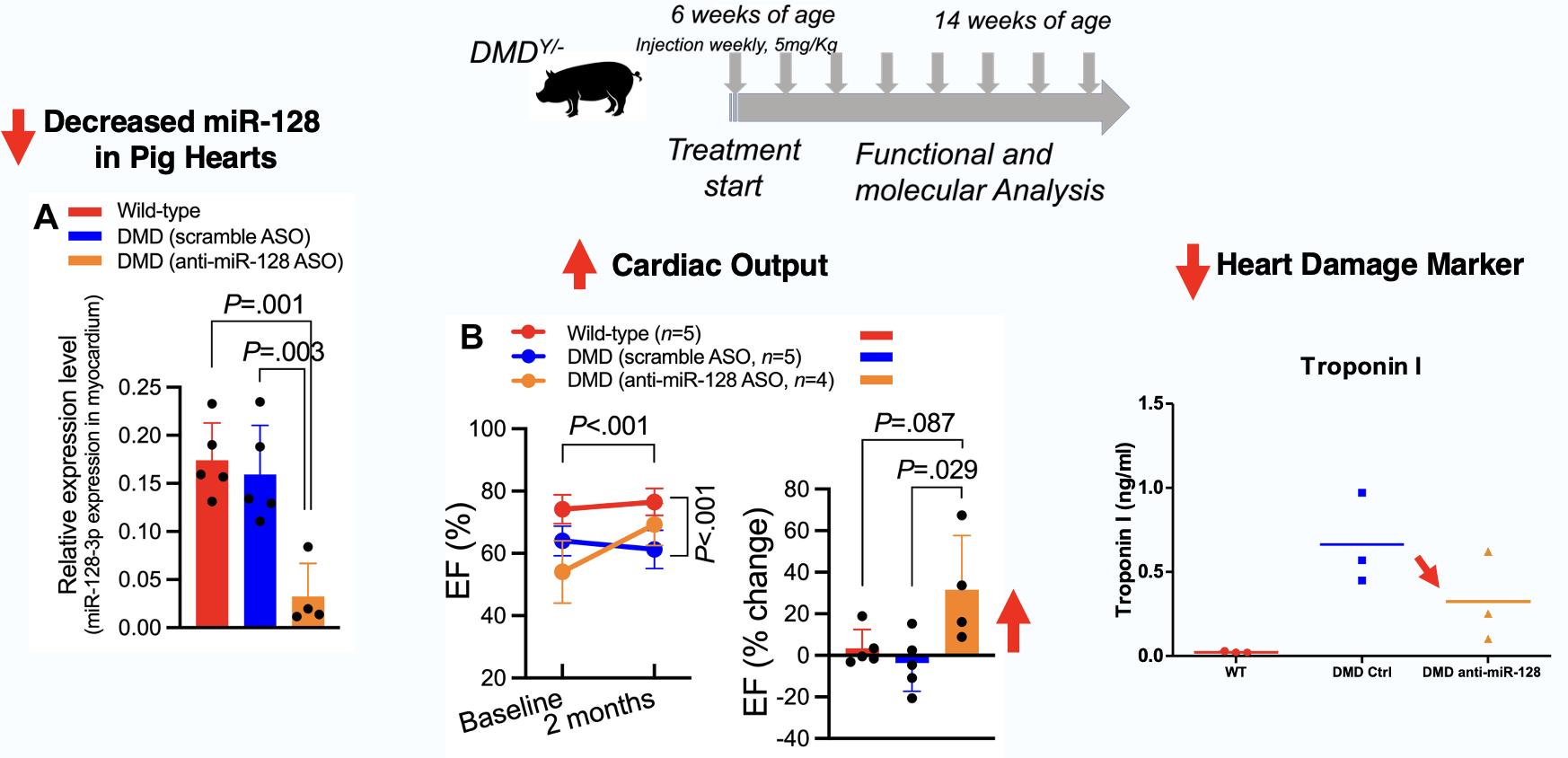
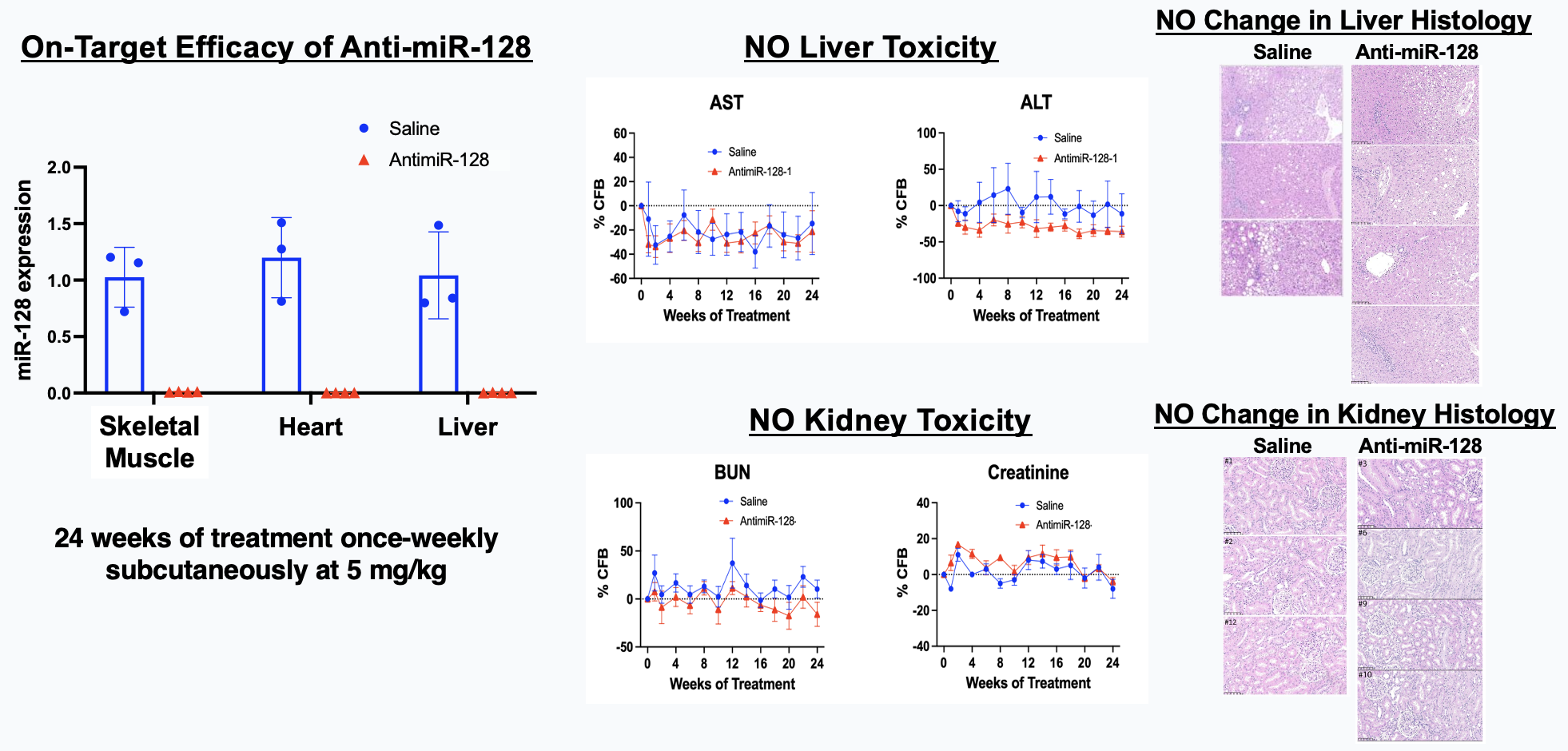
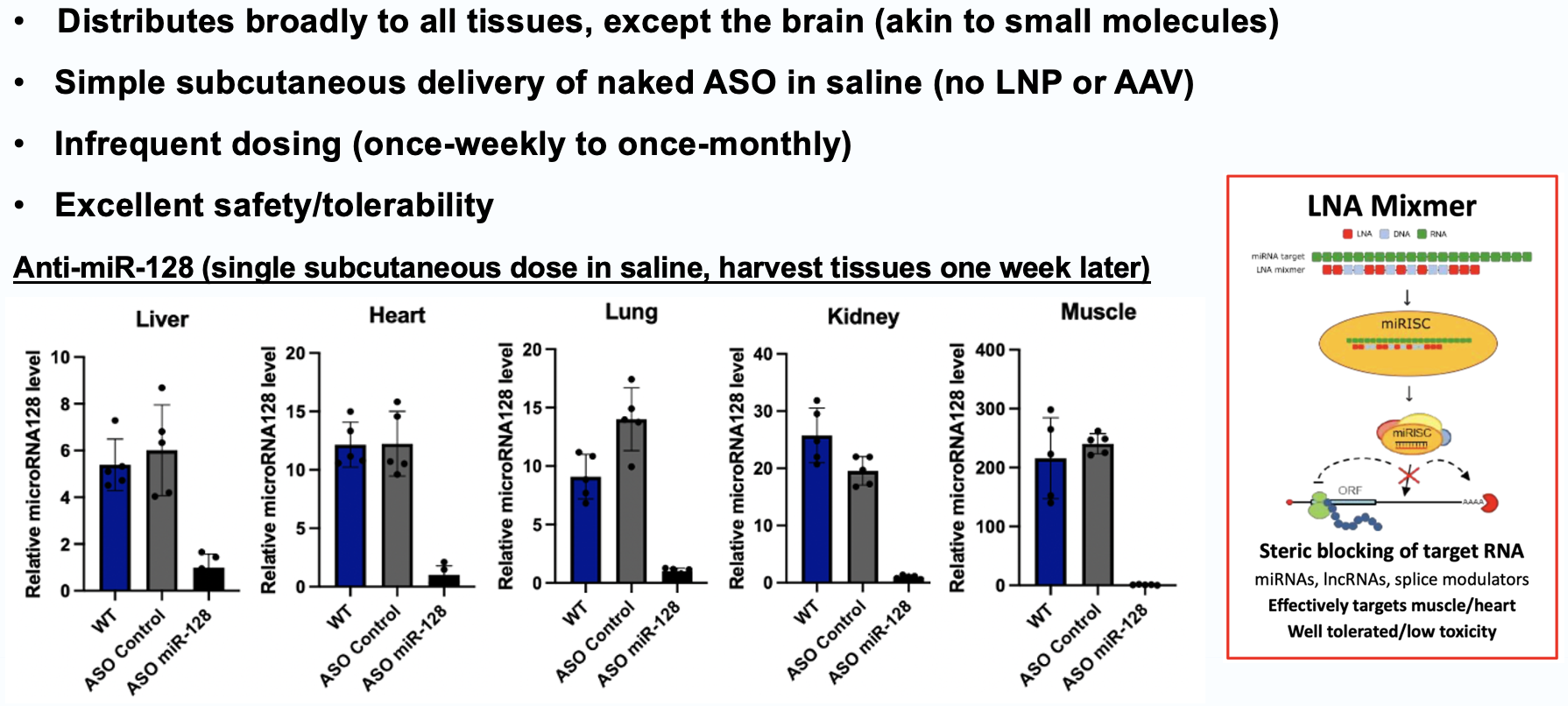
Elenae Therapeutics combines the native multi-organ biodistribution of LNA ASO mixmer chemistry with an AI-guided design engine to deliver potent, safe RNA medicines to tissues historically beyond the reach of oligonucleotide therapeutics.