Strategic Landscape
The RNA M&A Window Has Opened
Three major acquisitions totaling ~$15B in the last 18 months validate mixmer ASO chemistry, miRNA targeting, and muscle/cardiac delivery.
Cardior
$1.1B (Novo Nordisk)
- Cardiac miRNA (miR-132)
- LNA mixmer chemistry
Regulus
$1.7B (Novartis)
- Kidney miRNA (miR-17)
- 2'-MOE mixmer chemistry
Avidity
$12B (Novartis)
- Muscle/cardiac AOCs
- DMD + cardiac programs
- PMO chemistry
-
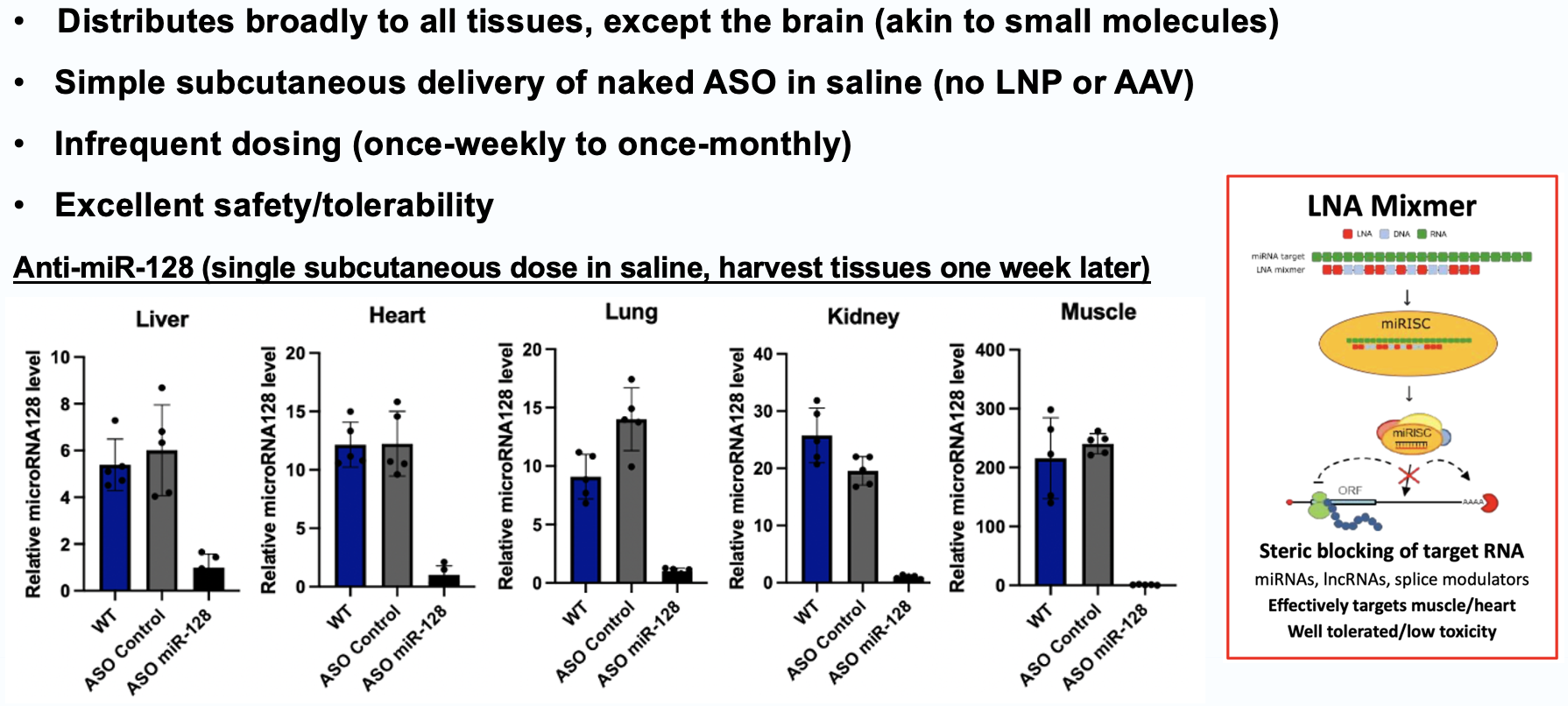
LNA mixmers de-risked by Cardior (Novo Nordisk), Santaris (Roche), miRagen
-
Cardiac + muscle delivery commanded premium valuation (Avidity–Novartis)
-
2026–2029 positioned as the next strategic acquisition window
The features defining these billion-dollar transactions—mixmer chemistry, reliable delivery to heart and muscle, true systemic reach—are the key strengths of Elenae’s scalable platform.